

AliveCor received FDA clearance for a new feature for its at-home EKG device, KardiaMobile 6L. Photo credit: AliveCor
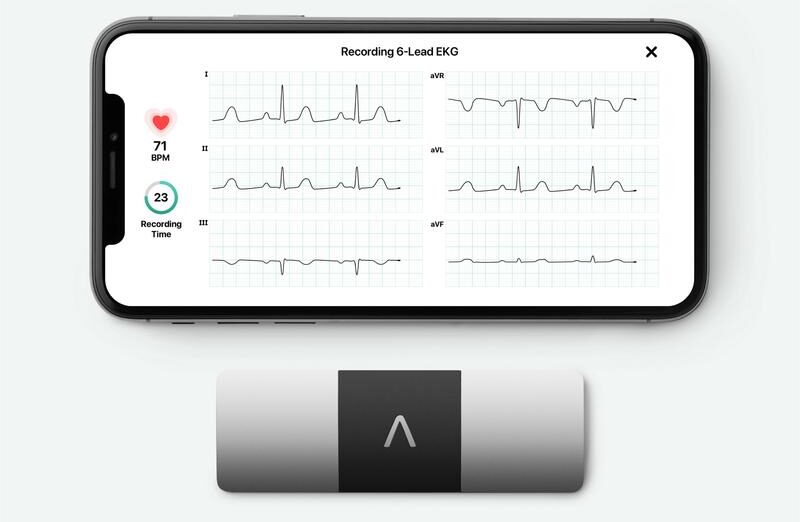
As more consumer-facing devices add features to measure the heart’s electrical activity, companies are looking for new ways to turn this data into valuable health information. AliveCor, a startup that makes at-home EKG devices, recently received 510(k) clearance for a feature that could help physicians detect if medications are affecting a patient’s QT interval.
The measurement tracks how long it takes for the heart’s ventricles to contract and relax. Sometimes, patients can develop a prolonged QT interval, which can increase their risk of developing a life-threatening arrhythmia. Certain medications can lead to QT prolongation, including some types of heart rhythm medications, antibiotics and antidepressants.
For example, if a patient is prescribed one of these medications and has other risk factors, a physician could use the device to monitor their QT interval.
Patients hold AliveCor’s KardiaMobile 6L device between their fingers for 30 seconds to get a reading. A physician can then access that data through a remote monitoring platform to manually measure a person’s QT interval. AliveCor also offers software to calculate QT intervals. Patients do not have access to the QTc values.
“Patient safety is paramount, and this is why we are proud to offer physicians the ability to monitor QTc through the convenience and quality of our device,” AliveCor CEO Priya Abani, said in a news release. “It is our hope that this important FDA clearance will help healthcare professionals identify and save patients from this potentially life-threatening condition.”
AliveCor isn’t the only company looking at this technology. An article recently published in Nature also tested the feasibility of using an Apple Watch for QT interval monitoring.
Photo Credit: Narongrit Doungmanee


